Table of Contents
What Is Hot Dip Galvanizing?
Hot dip galvanizing is a permanent corrosion protection process in which fabricated steel components are completely immersed in a bath of molten zinc maintained at approximately 450–460°C. Unlike surface coatings that simply adhere to steel, hot dip galvanizing creates a metallurgical bond between zinc and the steel substrate.
During immersion, molten zinc reacts with iron in the steel to form a series of zinc–iron alloy layers that grow outward from the steel surface. These alloy layers are significantly harder than the base steel itself and are topped with a layer of pure zinc. The result is a robust, abrasion-resistant coating that becomes an integral part of the steel rather than a separate film.
What makes hot dip galvanizing unique is its dual corrosion protection mechanism. First, the zinc coating acts as a physical barrier, isolating steel from moisture, oxygen, and corrosive elements.
Second, zinc provides cathodic (sacrificial) protection—meaning it corrodes preferentially to steel. Even if the coating is scratched or locally damaged, the surrounding zinc continues to protect the exposed steel, preventing rust from spreading.

With typical coating thicknesses ranging from 85 to 180 microns, hot dip galvanized steel can deliver a service life of 50 to over 100 years with little to no maintenance, making it one of the most reliable and economical corrosion protection systems available worldwide.
Why Hot Dip Galvanizing Is the Best Corrosion Protection
Hot dip galvanizing outperforms other coatings due to its unmatched durability and lifecycle cost advantages:
- Service life: 50–100+ years
- Complete coverage: Edges, welds, corners & hollow sections
- Self-healing: Zinc corrodes sacrificially
- Zero maintenance: No repainting required
How the Hot Dip Galvanizing Process Works (8 Steps)
The hot dip galvanizing process consists of eight precisely controlled stages, each critical to achieving a uniform, adherent, and long-lasting zinc coating. Any deviation in these steps can directly impact coating thickness, appearance, and corrosion performance.
1. Fabrication – Cutting, Welding & Assembly
All steel components are fully fabricated before galvanizing. This includes cutting, drilling, bending, welding, and assembly. Proper fabrication design is essential to allow free flow of molten zinc during dipping.
- Vent and drain holes are added to hollow sections to prevent air entrapment
- Welds must be continuous and free from slag or flux residues
- Overlapping surfaces should be avoided to prevent uncoated areas
2. Degreasing – Removal of Oils and Grease
Steel components are cleaned in an alkaline degreasing solution to remove oils, lubricants, paints, marking inks, and organic contaminants introduced during fabrication.
- Ensures acids can contact bare steel during pickling
- Prevents coating defects such as bare spots or black patches
- Typical solution: alkaline or caustic soda-based cleaners
Incomplete degreasing is a common cause of poor zinc adhesion.
3. Pickling – Removal of Rust and Mill Scale
After degreasing, steel is immersed in a pickling bath, usually containing hydrochloric acid, to dissolve rust (iron oxides) and mill scale formed during steel rolling.
- Produces a chemically clean and reactive steel surface
- Pickling time varies based on steel condition and thickness
- Inhibitors are added to minimize base metal attack
Proper pickling is essential for consistent zinc–iron alloy formation.
4. Rinsing – Acid Residue Removal
Following pickling, steel is thoroughly rinsed with clean water to remove residual acids and iron salts.
- Prevents contamination of the flux solution
- Reduces ash and dross formation in the zinc bath
- Improves overall coating appearance and uniformity
Multiple rinsing stages may be used in high-quality galvanizing lines.
5. Fluxing – Prevents Oxidation Before Dipping
Steel is immersed in a zinc ammonium chloride flux solution, which performs two critical functions:
- Removes any remaining oxides from the steel surface
- Prevents oxidation before immersion in molten zinc
Fluxing promotes rapid wetting of steel by molten zinc, ensuring uniform coating formation. Excess moisture is removed by air drying or pre-heating before dipping.
6. Galvanizing – Immersion in Molten Zinc
The prepared steel is slowly immersed into a bath of molten zinc maintained at 450–460°C.
- Zinc reacts metallurgically with iron to form zinc–iron alloy layers
- Coating thickness depends on steel chemistry, thickness, and immersion time
- Agitation and withdrawal speed influence coating smoothness
Typical immersion time: 3–15 minutes, depending on component size and thermal mass.
7. Cooling – Air or Water Quenching
After withdrawal from the zinc bath, the coated steel is cooled either by air cooling or water quenching.
- Solidifies the zinc coating
- Improves handling safety
- Optional passivation can be added during quenching to reduce white rust formation
Cooling method selection depends on product type and appearance requirements.
8. Inspection – Thickness & Adhesion Checks
The final step ensures compliance with ASTM A123, ISO 1461, or other specified standards.
- Visual inspection for coverage, runs, and surface defects
- Coating thickness measurement using magnetic gauges
- Adhesion testing to confirm no flaking or peeling
- Dimensional checks for fit and tolerance
Only components that pass inspection are approved for dispatch.
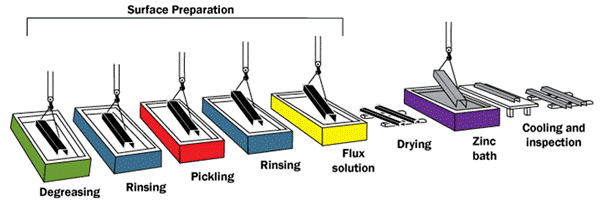
Pre-Galvanizing Surface Preparation (Most Critical Step)
Nearly 90% of hot dip galvanizing quality depends on proper surface preparation. Even the best galvanizing process cannot compensate for poor cleaning. Any oil, rust, mill scale, or oxide left on the steel surface will prevent zinc from bonding metallurgically, leading to coating defects such as bare spots, poor adhesion, or uneven thickness.
Process | Purpose | Chemicals Used |
Degreasing | Remove oils & lubricants | Alkaline solutions |
Pickling | Remove rust & mill scale | Hydrochloric acid |
Rinsing | Remove acid residues | Fresh water |
Fluxing | Prevent oxidation before dipping | Zinc ammonium chloride |
Let’s take a look at them in detail.
Degreasing – Removal of Oils and Organic Contaminants
Degreasing is the first cleaning step, designed to remove oils, greases, cutting fluids, shop dirt, and marking inks introduced during fabrication.
- Ensures acids can contact the steel surface during pickling
- Prevents coating defects such as black spots and bare areas
- Typically performed using hot or cold alkaline cleaning solutions
Incomplete degreasing is one of the most common causes of galvanizing failures.
Pickling – Removal of Rust and Mill Scale
After degreasing, steel is immersed in a hydrochloric acid pickling bath to dissolve rust (iron oxides) and mill scale formed during hot rolling.
- Exposes pure, reactive steel for zinc bonding
- Pickling time depends on steel condition and thickness
- Corrosion inhibitors are added to prevent over-pickling
Proper pickling is essential for consistent zinc–iron alloy layer formation.
Rinsing – Acid Residue Removal
Once pickling is complete, steel is thoroughly rinsed with fresh water to remove residual acid and dissolved iron salts.
- Prevents contamination of the flux bath
- Reduces ash and dross formation in the zinc kettle
- Improves coating uniformity and surface appearance
In high-quality galvanizing lines, multiple rinse stages may be used.
Fluxing – Oxidation Prevention Before Galvanizing
Fluxing is the final surface preparation step, using a zinc ammonium chloride solution.
- Removes any remaining microscopic oxides
- Prevents oxidation before immersion in molten zinc
- Promotes rapid and uniform wetting of steel by molten zinc
Steel is typically dried or preheated after fluxing to ensure moisture-free dipping.
Zinc Coating Formation & Thickness
When properly prepared steel is immersed in molten zinc, a metallurgical reaction occurs between iron (Fe) in the steel and zinc (Zn) in the bath. This reaction forms a series of zinc–iron alloy layers that grow outward from the steel surface, topped by a layer of pure zinc.
Unlike paint or sprayed coatings, these layers are bonded at the atomic level, making the coating exceptionally durable and abrasion resistant.
The coating structure consists of five distinct layers, each contributing to overall corrosion protection and mechanical strength.
Layer | Composition | Hardness (HB) |
Eta (η) | Pure Zinc | 100–150 |
Zeta (ζ) | Zinc-Iron Alloy | 200–250 |
Delta (δ) | Zinc-Iron Alloy | 300–400 |
Gamma (Γ) | Zinc-Iron Alloy | 400–500 |
Steel | Base Metal | — |
- Gamma, Delta, and Zeta layers form through diffusion of iron into molten zinc and provide exceptional hardness and wear resistance.
- The Eta layer of pure zinc serves as the primary sacrificial barrier, corroding preferentially to protect the underlying steel.
- The combined coating is typically harder than the base steel, making it resistant to mechanical damage during handling and installation.
Factors Influencing Coating Thickness
Zinc coating thickness is not arbitrary—it is influenced by several controlled and material-dependent factors:
- Steel thickness: Thicker steel retains heat longer, producing thicker coatings
- Steel chemistry: Higher silicon and phosphorus content increase coating growth
- Immersion time: Longer dwell times allow more alloy layer formation
- Bath temperature: Typically maintained at 450–460°C
- Surface condition: Properly cleaned steel promotes uniform layer formation
Typical Coating Thickness Ranges
Hot dip galvanizing produces coatings significantly thicker than most alternative systems:
- Small parts: 85–100 μm
- Medium structures: 100–130 μm
- Heavy beams & solar structures: 130–180 μm
These thickness levels enable hot dip galvanized steel to achieve 50–100+ years of corrosion protection, depending on environmental exposure.
Performance Advantage of Thick Zinc Coatings
Thicker zinc coatings directly translate to longer service life. Zinc corrodes slowly—typically 1–2 μm per year in most outdoor environments—making hot dip galvanizing one of the most reliable long-term corrosion protection systems available.
International Coating Thickness Standards
Hot dip galvanizing coating thickness is governed internationally by ASTM A123/A123M and ISO 1461, which define minimum zinc coating requirements to ensure consistent corrosion protection and long-term performance across different steel thicknesses and exposure conditions.
These standards specify minimum average coating thickness, testing procedures, and acceptable surface conditions for galvanized steel used in structural, industrial, and infrastructure applications.
Key Global Standards:
- ASTM A123/A123M
Widely used in North America and international projects, covering structural steel, plates, pipes, and fabricated assemblies. - ISO 1461
Commonly adopted across Europe, Asia, the Middle East, and global EPC projects for hot dip galvanized fabricated steel items.
Both standards are technically aligned and focus on durability, service life, and coating integrity.
Minimum Zinc Coating Requirements:
Steel Thickness | Outdoor Use | Indoor Use |
≤ 3 mm | 85 μm | 45 μm |
3–6 mm | 100 μm | 55 μm |
6–12 mm | 115 μm | 70 μm |
> 12 mm | 130 μm | 85 μm |
Important Notes:
- Outdoor requirements are higher due to continuous exposure to moisture, UV, and pollutants
- Indoor requirements assume controlled, low-corrosion environments
- Coating thickness generally increases with steel thickness due to thermal mass and reaction time
Coating Acceptance Criteria
- Coating thickness is evaluated as an average of multiple readings over a defined surface area
- Localized thinner spots may be acceptable if the overall average meets the standard
- Bare areas or uncoated surfaces beyond allowable limits are not permitted
Zinc Coating Measurement Methods
Several approved methods are used to verify compliance with ASTM and ISO standards:
- Magnetic thickness gauge (most common):
Non-destructive, fast, and widely accepted for field and plant inspection - XRF zinc weight measurement:
Determines coating mass per unit area, useful for audits and laboratory testing - Microscopy (cross-section analysis):
Highly accurate but destructive; used for detailed metallurgical evaluation - Gravimetric stripping test:
Measures zinc weight by chemically removing the coating; reference laboratory method
Why Standards Compliance Matters:
Meeting international coating thickness standards ensures:
- Predictable corrosion protection and service life
- Acceptance by project inspectors and EPC contractors
- Compatibility with infrastructure, renewable energy, and export requirements
Non-compliant coatings can lead to premature corrosion, project rejection, or costly rework.
Post-Galvanizing Inspection & Quality Control
Post-galvanizing inspection ensures that the zinc coating meets international standards, performs as intended, and is fit for service. Quality control is typically conducted in accordance with ASTM A123/A123M, ISO 1461, or project-specific specifications.
Inspection is carried out after cooling and before dispatch, ensuring defects are identified and corrected early.

Visual Inspection – Uniformity & Drainage
All galvanized surfaces are visually examined for:
- Complete coverage without bare steel
- Smooth, uniform appearance with proper zinc drainage
- Absence of excessive runs, spikes, or lumps that may affect fit or safety
Minor surface irregularities are acceptable if coating thickness and protection are not compromised.
Thickness Testing – Compliance with Standards
Coating thickness is measured using calibrated magnetic thickness gauges at multiple locations:
- Readings are averaged over defined surface areas
- Minimum thickness must meet ASTM / ISO requirements
- Extra attention is given to edges, corners, and welds
Thickness compliance is the primary acceptance criterion for galvanized coatings.
Adhesion Test – No Flaking or Peeling
Zinc coatings must be firmly bonded to the steel substrate.
- The coating should not flake, peel, or delaminate under normal handling
- Light impact or scraping should not cause separation
- Adhesion failures indicate improper surface preparation or process control
Bend Test – Crack-Free at 30° Bend
For applicable products, bend testing is performed to verify coating ductility.
- Steel is bent to approximately 30°
- The zinc coating must remain intact without cracking or peeling
- Minor hairline cracks in thick coatings may be acceptable if steel remains protected
Common Defects & Remedies:
Defect | Cause | Remedy |
Ash inclusions | Contaminated rinse or flux | Improve rinsing procedures |
Dross pimples | Zinc bath contamination | Regular kettle cleaning |
Bare spots | Incomplete pickling or degreasing | Improve surface preparation |
Zinc runs | Slow withdrawal or poor drainage | Increase withdrawal speed |
Most defects are process-related and can be corrected without affecting coating performance when identified early.
Optional Passivation Treatment
Optional passivation may be applied after galvanizing, particularly for components stored or transported before installation.
- Improves surface appearance during storage
- Reduces formation of white rust (zinc oxide)
- Does not affect coating thickness or long-term corrosion protection
Passivation is recommended for humid environments and long transit periods.
Industry Applications of Hot Dip Galvanizing
Hot dip galvanizing is widely used across industries where long service life, minimal maintenance, and consistent corrosion protection are critical. Its ability to protect complex steel fabrications makes it ideal for both infrastructure and industrial environments.
Construction & Infrastructure
Hot dip galvanizing is extensively used in infrastructure projects due to its long lifespan and reliability.
- Structural steel frameworks
- Highway guardrails and crash barriers
- Transmission towers and lighting poles
- Bridges, handrails, and walkways
Its maintenance-free nature reduces lifecycle costs for public and commercial projects.
Renewable Energy
Renewable energy installations demand corrosion protection for 25–30+ years in open, exposed environments.
- Solar mounting structures and trackers
- Wind turbine platforms and access systems
- Substation structures
Hot dip galvanized steel ensures consistent performance throughout the operational life of renewable assets.
Agriculture & Rural Infrastructure
Agricultural environments are highly corrosive due to moisture, fertilizers, and animal waste.
- Irrigation pipelines and water channels
- Farm fencing and gates
- Grain silos and storage systems
Galvanized steel withstands aggressive rural conditions with minimal upkeep.
Industrial & Manufacturing
Industrial facilities require coatings that resist abrasion, chemicals, and mechanical handling.
- Conveyor frames and material handling systems
- Machinery bases and support structures
- Platforms, ladders, and safety cages
The hard zinc–iron alloy layers provide superior wear resistance compared to painted systems.
Hot Dip Galvanizing vs Other Coatings
Coating Type | Lifespan | Maintenance |
Hot Dip Galvanizing | 50–100 years | None |
Epoxy Paint | 5–15 years | High |
Galvalume | 20–40 years | Low |
Powder Coating | 10–20 years | Medium |
Why Hot Dip Galvanizing Delivers the Lowest Lifetime Cost
While hot dip galvanizing may have a higher initial cost than paint, it offers significantly lower total ownership cost:
- No recurring repainting or shutdown costs
- Long service life even in harsh outdoor environments
- Reduced inspection and maintenance labor
- Predictable performance backed by global standards
Over the lifespan of a steel structure, hot dip galvanizing consistently outperforms alternative coatings in both durability and cost efficiency.
Conclusion
Hot dip galvanizing is one of the most proven and cost-effective corrosion protection methods for steel. By creating a permanent metallurgical bond between zinc and steel, it delivers long-lasting protection, complete coverage, and minimal maintenance.
With service lives of 50–100+ years, compliance with global standards, and reliable performance across construction, renewable energy, agriculture, and industrial applications, hot dip galvanizing consistently outperforms alternative coating systems. For projects that demand durability, safety, and long-term value, hot dip galvanizing remains the clear choice.





